Thao’s paper published in Biochemistry
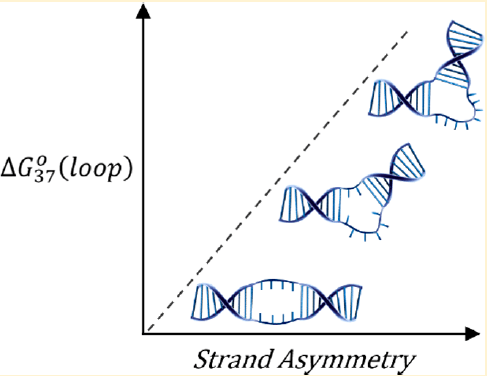
Internal loops within structured nucleic acids disrupt local base stacking and destabilize neighboring helical domains; however, these structural motifs also expand the conformational and functional capabilities of structured nucleic acids. Variations in the size, distribution of loop nucleotides on opposing strands (strand asymmetry), and sequence alter their biophysical properties. Here, the thermodynamics and structural flexibility of oligo-T-rich DNA internal loops were systematically investigated in terms of loop size and strand asymmetry. From optical melting experiments, a thermodynamic prediction model is proposed for the energetic penalty of internal loops that accounts for diminishing enthalpic and increasing entropic contributions due to loop size and strand asymmetry for bulges, asymmetric loops, and symmetric loops. These single-stranded domains become less sequence-dependent and more polymeric as the loop size increases. Single-molecule fluorescence resonance energy transfer studies reveal a gradual transition in conformation and structural flexibility from an elongated domain to an increasingly flexible bend that results from increasing strand asymmetry. The findings provide a framework for understanding the thermodynamic and conformational effects of internal loops for the rational design of functional DNA nanostructures.